 4 Evaluative Clinical Sciences, Odette Cancer Centre Research Program, Sunnybrook Research Institute, Toronto, Ontario, Canada Odette Cancer Centre, Sunnybrook Health Sciences Centre, Toronto, Ontario, Canada Department of Medicine, University of Toronto, Toronto, Ontario, Canada.3 Experimental Clinical Abdominal Oncology Unit, Istituto Nazionale Tumori-IRCCS-Fondazione G.2 Department of Oncology, Department of Public Health Sciences and Division of Cancer Care and Epidemiology, Queen's University, Kingston, Ontario, Canada.1 Evaluative Clinical Sciences, Odette Cancer Centre Research Program, Sunnybrook Research Institute, Toronto, Ontario, Canada.‖Determined by the study investigator by physical exam, sonography/MRI and/or biopsy. §Events after 2 consecutive missed disease assessments or initiation of post-surgery new anticancer therapy were censored at last disease assessment prior to the earlier date of ≥2 consecutive missed disease assessments and initiation of post-surgery new anticancer therapy if no events before new anticancer therapy, events were censored at last disease assessment before initiation of post-surgery new anticancer treatment. ‡EFS was defined as the time from randomization to the time of first documentation of disease progression that precludes definitive surgery, local or distant recurrence, a second primary cancer or death from any cause, whichever occurs first patients who did not experience an event at the time of data cutoff were censored at the date they were last known to be alive and event-free. †Hazard ratios (HR) and 95% CIs in the primary analysis and sensitivity analyses were based on a stratified Cox regression model analyses in subgroups were based on an unstratified Cox model. *Number of events/total number of patients (%). “Second breast malignancy” included in EFS definition “Positive margin at last surgery” and “second primary malignancy” removed from EFS definitionĥ. “Positive margin at last surgery” removed from EFS definitionĤ. “New anticancer therapy for metastatic disease” considered an EFS eventģ. This benefit was generally consistent across a broad selection of patient subgroups.Ģ. Conclusion: EFS sensitivity analyses show a robust treatment benefit of neoadjuvant pembro + chemo followed by adjuvant pembro for previously untreated non-metastatic TNBC. The benefit of neoadjuvant pembro + chemo followed by adjuvant pembro vs neoadjuvant chemo alone was generally consistent with the primary EFS results for all five sensitivity analyses and in each subgroup evaluated ( Table). 
Median follow-up was 39.1 months at the time of the Madata cutoff. Results: Among 1174 patients randomized, 784 were randomly assigned to the pembro + chemo group and 390 were randomly assigned to the placebo + chemo group. Treatment effects on EFS were examined in prespecified patient subgroups defined by nodal involvement (positive or negative), disease stage (II or III), menopausal status (pre-menopausal or post-menopausal), HER2 status (2+ by IHC but FISH- or 0-1+ by IHC), and LDH (>ULN or ≤ULN). Five prespecified sensitivity analyses for EFS were performed, including 2 that assessed the impact of different censoring rules and 3 that assessed the impact of different event definitions. Dual primary endpoints are pCR rate and EFS. 
Patients were stratified by nodal status (positive or negative), tumor size (T1/T2 or T3/T4), and carboplatin schedule (Q3W or QW). 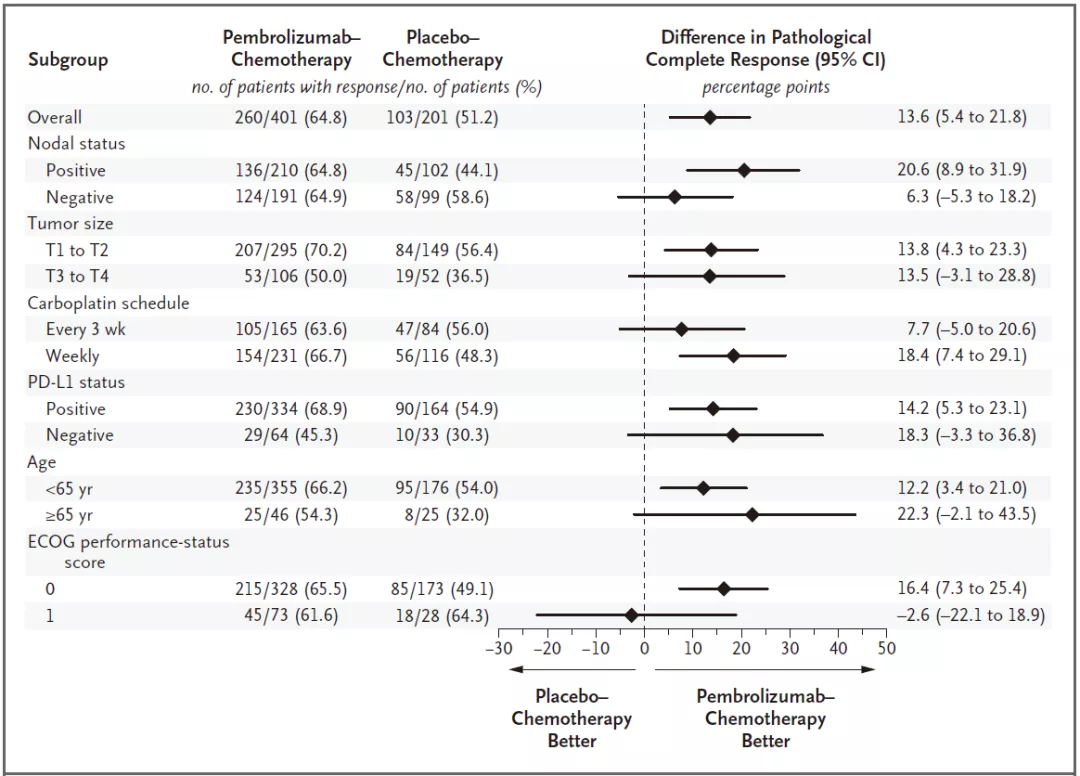
After definitive surgery, patients received pembro or placebo for 9 cycles or until recurrence or unacceptable toxicity (adjuvant phase). Methods: Patients with previously untreated, non-metastatic, centrally confirmed TNBC (stage T1c N1-2 or T2-4 N0-2 per AJCC) were randomized 2:1 to pembro 200 mg Q3W or placebo, both given with 4 cycles of paclitaxel + carboplatin, then with 4 cycles of doxorubicin or epirubicin + cyclophosphamide (neoadjuvant phase). To assess the robustness and consistency of the primary EFS result, prespecified sensitivity and subgroup analyses for EFS were performed. The primary analysis showed a statistically significant and clinically meaningful improvement in event-free survival (EFS) with pembro + chemo followed by pembro. Background: KEYNOTE-522 (NCT03036488) is a phase 3 study of neoadjuvant pembro + chemo vs placebo + chemo, followed by adjuvant pembro vs placebo in patients with early-stage TNBC. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
